Brady and Caretag partner for RFID-based surgical instrument tracking
Brady EMEA and Caretag have announced a strategic partnership to deliver an integrated solution for tracking surgical instruments in hospitals. By combining identification and UHF RFID technologies, the collaboration addresses regulatory requirements and operational challenges in sterile services and operating rooms across Europe.
Integrated approach to instrument traceability
With this partnership, Brady EMEA and Caretag introduce a combined solution for end-to-end management of surgical instruments. The system is designed to ensure that each instrument is clearly identified, reliably tracked, and fully documented throughout its lifecycle, from sterilization to use in the operating room and back.
The offering responds directly to increasing regulatory pressure, particularly the requirements of the EU Medical Device Regulation 2017/745. Hospitals must implement Unique Device Identification systems within defined transition periods up to 2027 and 2028. The joint solution supports compliance while aiming to improve operational efficiency and patient safety.
Addressing a complex hospital process
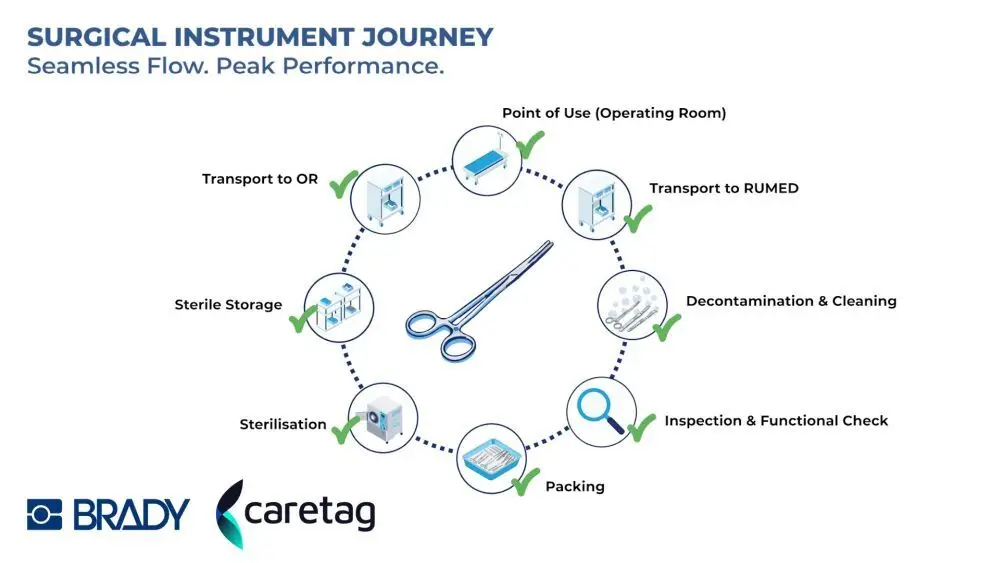
Instrument management remains one of the most critical and complex workflows in healthcare. Manual tracking methods are often time-consuming and prone to errors, creating risks in both clinical environments and regulatory compliance.
The collaboration between Brady EMEA and Caretag focuses on delivering consistent availability of the right instruments, supported by full documentation and reliable tracking. The goal is to reduce process risks while improving transparency across sterile services departments and surgical workflows.
Combining identification and RFID technology
The solution integrates complementary technologies from both companies. Brady contributes its patented data capture and identification systems, enabling reliable marking and reading of instruments across sterilization and surgical processes. This includes technologies for labeling, printing, laser marking, and engraving as part of a broader automatic identification and data capture approach.
Caretag provides its AI-supported UHF RFID platform and durable instrument tags. These tags are designed to withstand more than 2,500 sterilization cycles without affecting performance or instrument usability. The system allows simultaneous identification of multiple instruments, reducing packaging time by more than 33 percent compared to DataMatrix scanning.
In addition, the platform enables real-time location tracking, full usage history, and data-driven insights into instrument inventory. This supports hospitals in optimizing stock levels and reducing unnecessary procurement.
Relevance for healthcare operators and solution providers
For hospitals and sterile services departments, the integrated solution offers full lifecycle traceability and digital documentation aligned with MDR requirements. This can improve operational readiness, reduce instrument loss, and enhance patient safety.
For system integrators and solution providers, the partnership demonstrates how combining identification technologies with RFID platforms can create scalable, compliance-ready solutions for complex healthcare environments. The approach also highlights the growing role of data transparency and automation in clinical workflows.
Availability in Europe
The joint solution from Brady EMEA and Caretag is available to healthcare providers across Europe. It is positioned as a complete system for managing surgical instruments with a focus on traceability, compliance, and operational efficiency.
Talk to Brady
Ready to make surgical instrument tracking faster, safer, and fully traceable?
Brady EMEA can help you explore how RFID-based identification and Caretag’s platform can support your hospital, sterile services department, or healthcare project. Contact Brady EMEA to learn more, request a demo, or discuss your traceability requirements.

